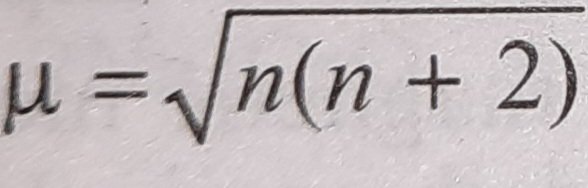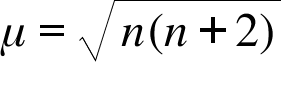58. If spin only magnetic moment of vcl, is 1.73 BM then correct formula is :- * vci, (2) VCI, X (3) VCI, (4) VCI, 53 -1.732. 3ds socarz?
56.Spin only magnetic moment of Mnx+ ion is root 15B.M.Then what is tge value of X OPTIONS:A)6 B)4 C)2 D)8
20. which of following have almost equal spin only and observed magnetic moment a)Fe(II) b)Ni(II) c)Cu(II) d)V(II)

The highest value of the calculated spin-only magnetic moment (in BM) among all the transition - YouTube

Write the spin only formula and give the unit of magnetic moment - Chemistry - Structure of Atom - 15663443 | Meritnation.com
![SOLVED: The spin-only formula to calculate the magnetic moment of a compound is: p = [N(N+2)]^(1/2) * uB where N is the number of unpaired electrons and uB is the Bohr magneton. SOLVED: The spin-only formula to calculate the magnetic moment of a compound is: p = [N(N+2)]^(1/2) * uB where N is the number of unpaired electrons and uB is the Bohr magneton.](https://cdn.numerade.com/ask_images/8bd8e65e68104e63897b868130353014.jpg)


![The magnetic moment (spin only) of [NiCl4]2– is - askIITians The magnetic moment (spin only) of [NiCl4]2– is - askIITians](https://files.askiitians.com/cdn1/cms-content/common/www.askiitians.comonlinetestforumsimages204-1316_sataug1615-54-35.jpg.jpg)


![Assamese] What is 'spin only' formula? Assamese] What is 'spin only' formula?](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/8950532.webp)
![The spin-only magnetic moments of k\u0104[fe(oxalate),] and k,[ru(oxalate),] are The spin-only magnetic moments of k\u0104[fe(oxalate),] and k,[ru(oxalate),] are](https://cdn.eduncle.com/library/scoop-files/2020/7/image_1594373088294.jpg)